Fresh water is an important and widespread natural resource. Microorganisms play key roles in freshwater biogeochemical processes, which are mostly studied during summer. However, with more than half of Earth's freshwater bodies subject to freezing, a complete picture of freshwater ecosystem processes necessitates study of ice-covered periods. Despite the importance of microorganisms and the widespread nature of ice covers, little is known about the mechanisms by which microorganisms interact with ice or the biosignatures they may impart to the freshwater ice matrix. The goals of this research are to determine whether microorganisms and metabolites can be detected in lake ice, how they differ from those in the water column, and how microbial life in Lake Superior and the Keweenaw Waterway survives over winter.
The aim is to assist a small community with a population less than 10,000 to improve nutrient removal of ammonia, nitrogen, and phosphorus from the wastewater treatment lagoons using innovative technologies.
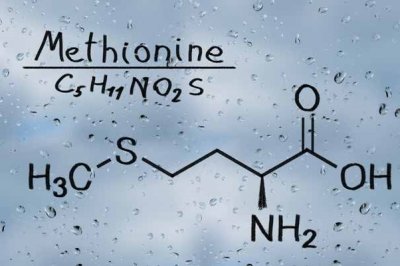
Abiotic fate of dissolved free amino acids considerably contributes to the cycling of dissolved sulfur and nitrogen in natural aquatic environments. The fate of these compounds present in fresh and saltwater environments is poorly understood. Some organic molecules, including amino acids, are taken up and reused by organisms living in the water, but a significant proportion is broken down through chemical and physical processes and these smaller degradation products enter a complex cycle of nitrogen and sulphur containing molecules. The photochemical process is one important degradation pathway.
Theoretically, the energy from sunshine could degrade certain amino acids, making them unavailable for living creatures. However, there is evidence that dissolved organic molecules generate very short-lived reactive intermediates under sunlight irradiation and these reactive intermediates degrade the amino acids – the photosensitising effect of dissolved organic molecules.
Dr Daisuke Minakata and his research group were prompted to develop a theory to explain the loss of critical amino acids from water. He assumed that other organic molecules absorb the energy carried by the rays of sunshine and those reactive intermediates transform amino acids into small pieces. According to his theory, such light-activated compounds could act as catalysts, inducing the indirect breakdown of critical amino acids.
This work is funded by the National Science Foundation, award number 1808052.
Publications
Barrios, Benjamin.; Minakata, D. Aqueous-phase single-electron transfer calculations for carbonate radicals using the validated Marcus theory. Environmental Science & Technology Letters. 2023. DOI: 10.1021/acs.estlett.2c00913.
B Barrios, B Mohrhardt, P Doskey, D Minakata, Mechanistic insight into the reactivities of aqueous-phase singlet oxygen with organic compounds, Environmental Science & Technology, 2021, 55(12), 8054–8067. DOI: https://doi.org/10.1021/acs. est.1c01712
Image Gallery