A new theory about virus surfaces—that they're hydrophobic—has opened up new processes to improve vaccine production, potentially making them more affordable around the world.
Virus Flocculation
Virus hydrophobicity is the focus of research by Caryn Heldt, an associate professor of chemical engineering, who won a National Science Foundation (NSF) Early Career Award grant. Heldt's CAREER Award will help her explore a process that causes viruses to clump, making them easier to remove.
This is a completely different approach to virus removal. And it builds off a new theory about virus surface chemistry. In her past research, Heldt delved into virus hydrophobicity. That is, how virus surfaces repel water. In the presence of water-loving osmolytes, viruses tend to stick together, making them easier to catch through filtration membranes.
Heldt explains how osmolytes and viruses interact in a solution in her TL;DR video up in the story header (that's too long; didn't read, folks.)
In a nutshell: Basically, viruses are hydrophobic, keeping a layer of water molecules surrounding them at a distance. The osmolytes pull away that layer. Once that happens, those hydrophobic surfaces are attracted to each other, causing the viruses to clump. Heldt and her team can then catch the virus clumps with larger-pore—and less expensive—screens to remove protein and virus impurities.
Watch the video below or catch up on the live tweeted chat with Heldt from March.
Caryn Heldt: Virus Flocculation Career Award
Live Chat via Storify
To Purify A Virus: #NSFCareer Researcher Caryn Heldt
Purifying a virus may sound counterintuitive. But improving virus purification could make vaccines easier to produce and more accessible around the world. Chemical engineer Caryn Heldt's work, funded by #NSFcareer, builds on new theories about virus chemistry to purify or remove them more easily.
Live chat with @carynheldt starts in 10 minutes. Talking #virus science #mtulive (image from @splette #scistyle) pic.twitter.com/dC9dVn5T8h
— MTU Research (@mturesearch) March 24, 2016
Enter the Challenge
Removing viruses is not easy to do. They're small; they're more chemically inert than bacteria and many molecules; they're highly variable in terms of size, shape, and surfaces. But they all share something in common: a new line of virus research suggests that virus surfaces are hydrophobic (water-fearing). That hydrophobicity could help overcome the challenges of virus removal, making them easier to purify.
@carynheldt Most people hear “virus purification” and have a ???? moment. Why do we need to purify viruses? #mtulive #BasicScience
— MTU Research (@mturesearch) March 24, 2016
Why purify a #virus? Join @carynheldt Thurs 2 p.m. EDT to find out. #mtulive #NSFcareer #MichiganTech pic.twitter.com/MuSJlAHGak
— MTU Research (@mturesearch) March 23, 2016
@mturesearch Our focus for purification is for improved vaccine manufacturing. Reducing cost could increase adaption around the world.
— Caryn Heldt (@carynheldt) March 24, 2016
Meet the Solution
To ease global access to vaccines, we need a solution--literally, a liquid solution of water full of viruses, a smattering of proteins, and some molecules called osmolytes. These, and a porous membrane used for filtration, are the key players in Heldt's research.
@mturesearch #MTULive I really like this picture from #MichiganTech illustrator Jordan Blahnik pic.twitter.com/JYCXynig2d
— Caryn Heldt (@carynheldt) March 24, 2016
@mturesearch This will take several tweets...Osmolytes help to maintain water in the cells of deep sea creatures, living in high salt.
— Caryn Heldt (@carynheldt) March 24, 2016
@mturesearch #MTULive Osmolytes also help to stabilize proteins. They are often in therapeutic protein formulations.
— Caryn Heldt (@carynheldt) March 24, 2016
@mturesearch #MTULive Using osmolytes in manufacturing is a great way to control water with compounds that are already FDA approved.
— Caryn Heldt (@carynheldt) March 24, 2016
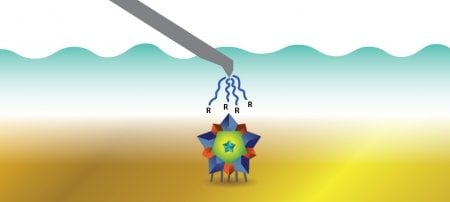
Osmolytes can be sugars, proteins, or other molecules. Heldt found in her research that glycine, an amino acid, and mannitol, a sugar, can be used to remove viruses. Here's how:
@carynheldt Ok, so we’ve got a #virus and its buddies with some osmolytes in solution. What happens? #mtulive
— MTU Research (@mturesearch) March 24, 2016
@mturesearch #MTULive The picture shows how osmolytes can remove water from the virus because osmolytes bind or cage water.
— Caryn Heldt (@carynheldt) March 24, 2016
@mturesearch #MTULive Viruses are hydrophobic, so without water, they aggregate. This allows for quick and easy purification.
— Caryn Heldt (@carynheldt) March 24, 2016
@mturesearch #MTULive Hydrophobicity means water fearing. So it is easy to take water from a surface that doesn't like it much.
— Caryn Heldt (@carynheldt) March 24, 2016
Osmolytes cause viruses to clump together, or flocculate. “And we if flocculate the viruses then we can use larger pore- size filters to remove them,” she says. Bigger pores = less expensive material = streamlined vaccine production
Cover a Virus or Two
Hydrophobicity helps explain why Heldt's process using osmolytes causes viruses to clump. The next stage of her research will focus on figuring out the specific chemical and physical features of different viruses. While there is a lot of variability of viruses, surface hydrophobicity may be a shared feature.
@mturesearch #MTULive We want to purify the two types of virus, enveloped and non-enveloped. The goal is a process that is universal.
— Caryn Heldt (@carynheldt) March 24, 2016
@mturesearch #MTULive For non-enveloped virus, we use porcine parvovirus. It is small and chemically stable. pic.twitter.com/Plj6N8WAlE
— Caryn Heldt (@carynheldt) March 24, 2016
@mturesearch #MTULive Our enveloped model is Sindbis virus. Also small and stable. pic.twitter.com/aoVctAxU1H
— Caryn Heldt (@carynheldt) March 24, 2016
@carynheldt Why both enveloped and non-enveloped? #mtulive #virus
— MTU Research (@mturesearch) March 24, 2016
@mturesearch #MTULive We would like to find a universal way to purify most vaccines. So, we need to use both types of virus.
— Caryn Heldt (@carynheldt) March 24, 2016
Wrap up
Final question @carynheldt, why study #virus purification? #mtulive #nsfcareer
— MTU Research (@mturesearch) March 24, 2016
@mturesearch #MTULive My mom and sister are nurses. I want to contribute to health sciences without having to gain a bedside manner.
— Caryn Heldt (@carynheldt) March 24, 2016
.@mturesearch what can we expect from the next #MTULive discussion about #science at @michigantech?
— Science MI (@sciencemi) March 29, 2016
Michigan Technological University is an R1 public research university founded in 1885 in Houghton, and is home to nearly 7,500 students from more than 60 countries around the world. Consistently ranked among the best universities in the country for return on investment, Michigan's flagship technological university offers more than 185 undergraduate and graduate degree programs in science and technology, engineering, computing, forestry, business, health professions, humanities, mathematics, social sciences, and the arts. The rural campus is situated just miles from Lake Superior in Michigan's Upper Peninsula, offering year-round opportunities for outdoor adventure.




Comments