The Bhakta Rath Research Award honors a graduate student and faculty mentor for in-depth work with social impact. The 2019 winners are two biomedical engineers with a sticky past.
A smart adhesive doesn’t adhere all the time. In 2015, when Ameya Narkar started his doctoral research with Bruce Lee, associate professor of biomedical engineering at Michigan Technological University, the two turned to biological sources for a glue that could be turned on and off.
They started with an amino acid found in mussel foot proteins called DOPA (3,4-dihydroxyphenylalanine). Yes, that’s a mussel like the ones found on piers and boat bottoms, and yes, DOPA is related to the more-familiar chemical dopamine. DOPA helps mussels cling to their underwater homes and, in 2016, Lee earned a Young Investigator Program (YIP) award from the Office of Naval Research to explore its potential for underwater adhesives.
Together, Narkar and Lee explored more possibilities: easy-to-remove bandages, customized medical devices and implants, glue for cameras to attach to fish and other underwater environmental monitoring. They say the key to their research success is early mornings — they’re both in the lab by 8 a.m. — and finding the positives even when the initial data seems gloomy.
Here are more insights into the hard work and thoughtful process of Narkar (AN) and Lee (BL).
Q: What’s your project about?
AN: We study smart adhesives. This isn’t a stick-peel-and-done kind of glue like you’d find on a regular Band-Aid.
BL: Yes, a smart adhesive can be turned on and off depending on its environment. We specifically worked on a design that uses changes in pH to make our material become more adhesive and then less adhesive.
In biomedical engineering, we work on many different fronts. We develop and test materials that need to be characterized in order to be biocompatible, and we need to understand the chemical, mechanical and biological systems we work with. To create a smart adhesive, the work is not as simple as making a material sticky and not sticky anywhere at any time; it’s about designing a device that works within specific environments for specific reasons. Our job is to figure out how and why.
Q: Where did you get the idea?
AN: Well, I have kinda hairy arms. And ripping off a Band-Aid is pretty painful. We wondered what would it be like if you could remove a bandage without any pain?
BL: I have been working with DOPA and other synthetic versions of mussel adhesive proteins since I was a graduate student more than 20 years ago. Under certain conditions the adhesive properties of DOPA can be significantly reduced and this process is often irreversible. This is typically undesirable because you want the adhesive to be sticky. Ameya designed the adhesive so that its adhesion chemistry could be reversed.
"I’m extremely proud of the work of this team. Being able to manipulate the adhesion of a material makes it a ‘smart’ material; and these have applications not only in the medical field but far beyond."
Q: How have your methods helped make the project successful?
AN: While the specifics get complex, we generally focus on reduction and oxidation reactions. That is, chemical shifts that change how a material interacts with its environment — the classic example is how rust forms when exposed to water.
We used pH to reduce and oxidize our smart adhesive, effectively turning it on and off. Our experimental design involved adding a boronic acid to the adhesive network, and this acid was able to form reversible bonds with DOPA that were responsive to pH. DOPA is adhesive in its reduced state. Once it becomes oxidized, once the pH is elevated, DOPA changes its chemical structure and physical properties — it’s not really adhesive anymore.
On its own, DOPA can’t go back to the reduced form either. Incorporating the boronic acid meant that if we brought up the pH and made the environment basic, then the boronic acid formed a protective coordinate complex with the DOPA so it wasn’t oxidized and wasn’t adhesive. When the pH was lowered, being more acidic, the bonds were broken and DOPA could become fully adhesive again. To do this we had to figure out all the details of how the boronic acid compound and DOPA interact as well as the physical and chemical limits of the reactions.
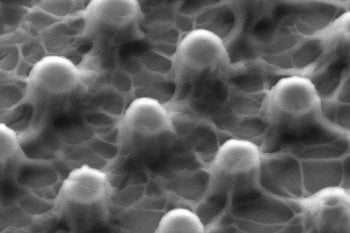
We then took it another step up. We coated the boronic-DOPA compound on micropillars, which increases the surface area and helps us adjust and control adhesion.
And we have the support to make everything happen. Our biomedical engineering department is full of approachable experts. It’s a small team and an effective one. I could walk down to a faculty member’s office and ask for advice when our project branched into areas beyond our lab’s expertise. Plus, I was able to work closely with the people in the Applied Chemical and Morphological Analysis Laboratory and the microfabrication facility. Collaboration is essential to successful research.
"I joined the lab as a very young investigator with almost no research background at all. But Lee trained me to develop a deep scientific outlook and he also inspired me to cultivate an excellent work ethic. He also always encouraged me to look at the positive side, as most days in research are not eureka days."
Q: What are the challenges you face in this field?
BL: Using pH is nice because it is easier to control in a laboratory setting. However, it is highly impractical. What we are currently working on is to use electricity to tune the adhesive property. In the future we envision that we could simply push a button and the adhesive could be turned off. Ameya’s results served as an initial first step in understanding adhesion chemistry. We are building off of his results to create an electro-responsive adhesive.
AN: The next steps in the research focus on the next gen of smart adhesives that are electro-responsive, so you run a small current through it and it changes from strong to weak adhesion almost instantaneously.
Q: Who benefits from your smart adhesives research?
AN: One of the motivating factors right from the beginning has been designing an adhesive that can form a very strong bond when desired but on the flip side is easily detachable and painless. Versatility makes it appealing to many end users; we’re figuring out the mechanisms that make it happen and there’s still a lot of work to be done.
BL: Adhesives that provide strong yet temporary wet adhesion can be used to anchor and release underwater sensors and devices, and increase the mobility of automated or unmanned underwater vehicles or robots. Additionally, smart bioadhesives can be integrated with wearable sensors, used to create painlessly removable wound dressings or used as temporary adhesives for fixation of prosthesis and implantable medical devices.
"I try to give my students freedom to explore and even make mistakes. I usually let them decide how they should plan their experiments. I also set high expectations and I always challenge them to do more. If the student is willing to improve and grow, he or she will become an independent researcher that can think on his or her own."
Q: Why do you care — what motivates you?
AN: It is the curiosity. You start out reading an article related to your work, then you reflect on it, then you start thinking, “Well, what if I tried this; what if I change this?” It’s all the what-if questions. And since we’re lucky enough to have all the resources we need at Michigan Tech, we could do any type of characterization technique we could think of, so we could do the experiment right away. It’s basic curiosity to want to know what is happening.
BL: I am interested in solving problems, especially those that are challenging. Even though I have worked on using DOPA to design adhesives for over 20 years, I still find unique aspects and unknown aspects of the mussel adhesive chemistry that we could potentially incorporate to design better biomaterials and medical devices.
"How can DOPA-containing polymers and proteins create both [internal and external] bonding at the same time? The answer is far from clear, yet nature seems to use only one molecule for two different things. Here is where Ameya and Bruce have, in my opinion, made their biggest impact. They have delved into the fundamental relations between DOPA chemistry and adhesion."
Michigan Technological University is an R1 public research university founded in 1885 in Houghton, and is home to nearly 7,500 students from more than 60 countries around the world. Consistently ranked among the best universities in the country for return on investment, Michigan's flagship technological university offers more than 185 undergraduate and graduate degree programs in science and technology, engineering, computing, forestry, business, health professions, humanities, mathematics, social sciences, and the arts. The rural campus is situated just miles from Lake Superior in Michigan's Upper Peninsula, offering year-round opportunities for outdoor adventure.






Comments